Data Integrity Solutions
Ensuring Regulatory Compliance & Digital Trust

Ensuring Regulatory Compliance & Digital Trust
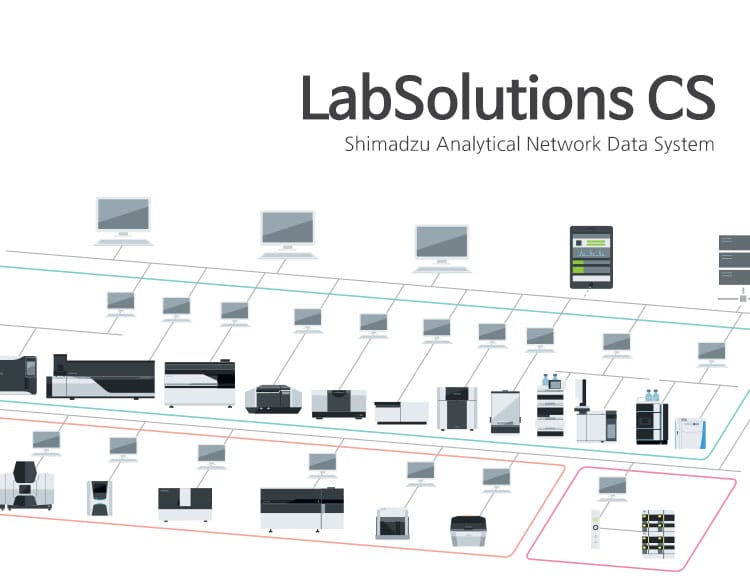
Pacific Group provides advanced Data Integrity Solutions designed to meet international regulatory standards such as USFDA 21 CFR Part 11 and ISO 17025. These solutions ensure secure, traceable, and compliant handling of electronic records and signatures, safeguarding operational transparency and regulatory readiness.
Deployed across more than nine major industries in Nepal, Pacific’s data integrity systems help businesses and labs maintain audit trails, prevent data manipulation, and align with global quality assurance frameworks.